Implanted heart monitor3/4/2023 It is a miniature percutaneous LAP sensor that is robust, wireless, and leadless, and includes a novel drift compensation mechanism. The V-LAP system (Vectorious Medical Technologies, Tel Aviv, Israel) is a next generation of implantable LAP monitoring systems, and uses advanced application-specific integrated circuit–based technologies.Further evaluation of this system is required. However, when the results were analyzed using the CHAMPION trial endpoint of recurrent HF hospitalizations, the results of the LAPTOP-HF trial were similar to those of the CHAMPION trial. The overall result for this trial was negative, demonstrating no reduction in a combined endpoint of recurrent HF hospitalizations and complications of HF therapy. Enrollment in the LAPTOP-HF trial was stopped early, due to a perceived excess of implant-related complications. Jude Medical/Savacor, Inc.), a system that allowed for direct measurement of LAP in patients with ambulatory HF, was studied in a prospective randomized controlled outcomes study, the LAPTOP-HF (Left Atrial Pressure Monitoring to Optimize HF Therapy) trial. Also currently under development are devices to monitor left atrial pressure (LAP) because LAP as a direct reflection of left ventricular filling pressure is the primary pressure target for the management of HF, direct measurement of LAP may potentially provide more clinical information in the management of HF than measurement of right-sided pressures or PAPs.Another system (developed by Endotronix, Inc., Woodridge, IL) appears to be similar to the CardioMEMS HF System, except for a different external user interface. This Medtronic device monitors not only PAP, but also cardiac arrhythmias, patient activity, and heart rate. Other PAP measurement systems are in development, including a small, implanted sensor that has a battery in the capsule and talks through intrabody communication to a Reveal LINQ Insertable Cardiac Monitor device co-implanted in the patient.The reported real-world experience with this system has also been encouraging. Device complications were remarkably low, with freedom from device-related or system-related complications of 98.6%, and overall freedom from pressure-sensor failures of 100%. 
Other key findings include a significant reduction in PAP, a significant increase in the number of days alive and out of the hospital for HF, a significant reduction in the proportion of patients hospitalized for HF, and a significant improvement in quality of life in treatment versus control patients. The Chronicle IHM (Medtronic, Inc., Minneapolis, MN) evaluated this device reported a 57% reduction (p 17 months. The first major breakthrough in the implantable monitoring device arena was a right ventricular sensor, which, by measuring pressure during pulmonic valve opening, could estimate the pulmonary artery diastolic pressure.Similarly, serum B-type natriuretic peptide has yet to be proven helpful in the ongoing management of patients with chronic HF. For example, daily measurement of body weight has a sensitivity of only 9% for the development of a new HF exacerbation. 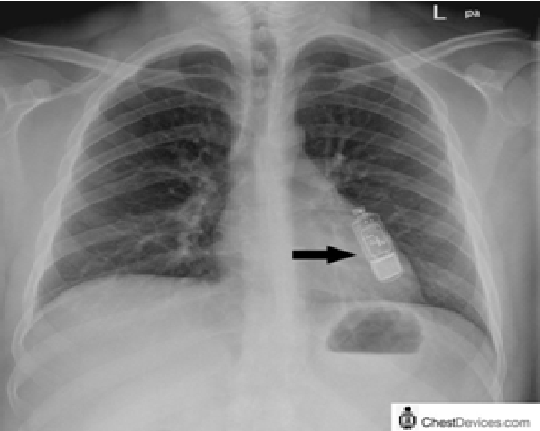
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
